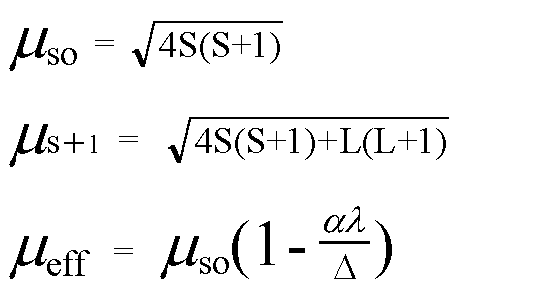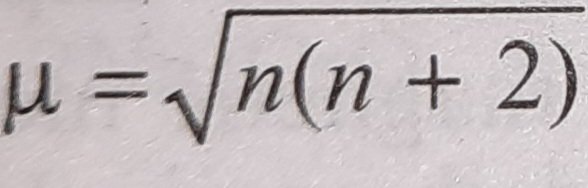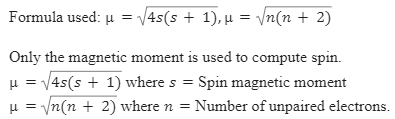![SOLVED: The spin only formula to calculate the magnetic moment of compound is p = [N(N+21]4/2 u8 number of unpaired electron and UlB is the Bohr magneton compound ZnCO CuSOs FeSO4 CoCh SOLVED: The spin only formula to calculate the magnetic moment of compound is p = [N(N+21]4/2 u8 number of unpaired electron and UlB is the Bohr magneton compound ZnCO CuSOs FeSO4 CoCh](https://cdn.numerade.com/ask_images/8bd8e65e68104e63897b868130353014.jpg)
SOLVED: The spin only formula to calculate the magnetic moment of compound is p = [N(N+21]4/2 u8 number of unpaired electron and UlB is the Bohr magneton compound ZnCO CuSOs FeSO4 CoCh

The highest value of the calculated spin-only magnetic moment (in BM) among all the transition metal complexes is :











![The magnetic moment (spin only) of [NiCl4]2– is - askIITians The magnetic moment (spin only) of [NiCl4]2– is - askIITians](https://files.askiitians.com/cdn1/cms-content/common/www.askiitians.comonlinetestforumsimages204-1316_sataug1615-54-35.jpg.jpg)